Strategic intelligence, built for Medical Affairs
One natural-language brief → 60+ pages of board-ready Medical Affairs strategy in a single session. Not a chatbot. Not a search tool.
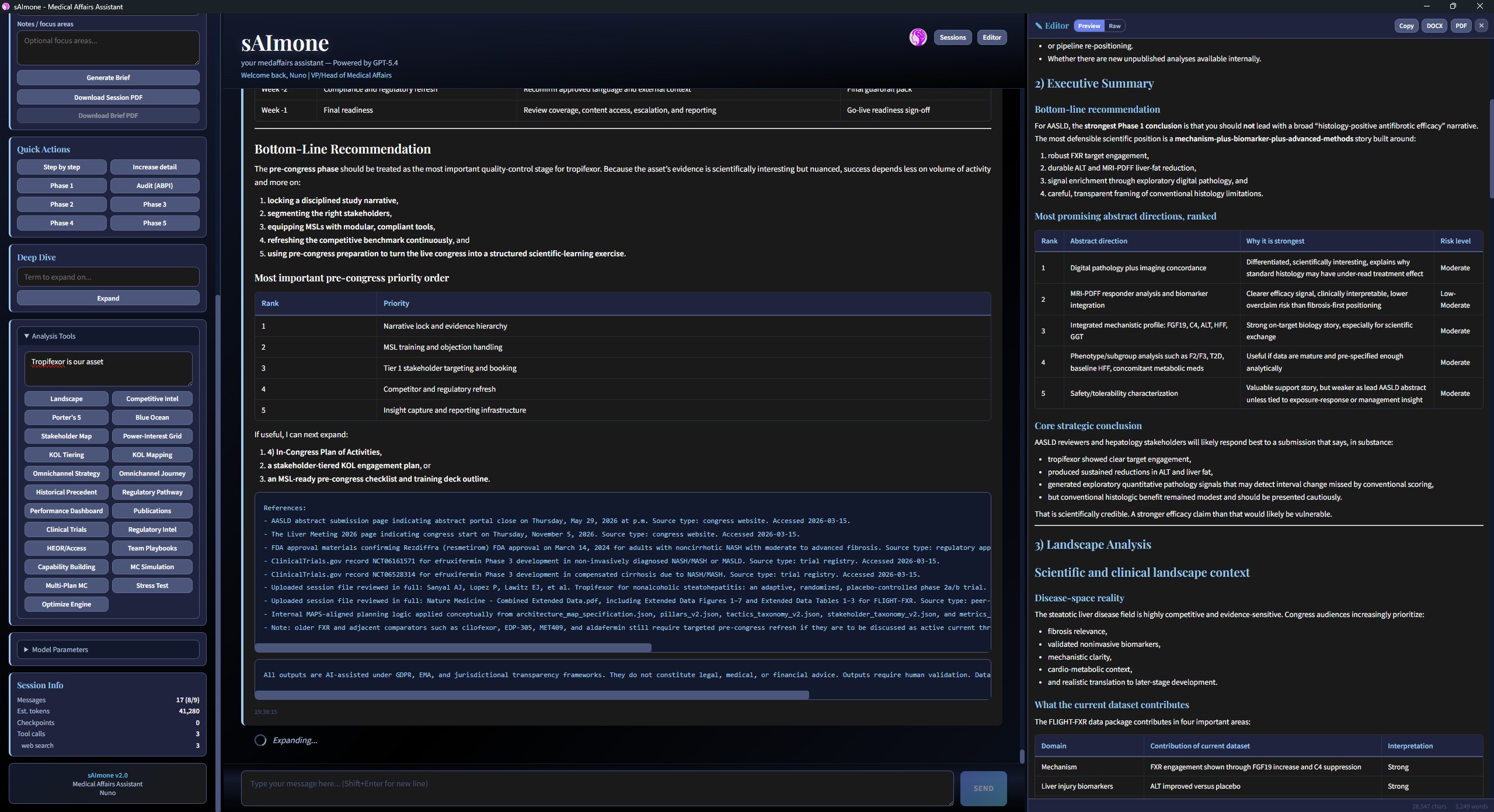
Competitive landscape analysis
Pipeline tracking, evidence mapping, and competitive positioning across therapeutic areas. A similarity engine identifies strategic parallels between compounds, mechanisms, and market dynamics.
Evidence synthesis
Structured analysis across clinical trials, publications, congress abstracts, and regulatory filings — with full citations. Extracts endpoints, hazard ratios, and p-values directly from source data.
One session, five deliverables
Strategic Summary for leadership, Operational Handbook for field teams, Medical Writer Handoff, Compliance Extract with evidence grading, and MSL Field Guide — all traceable to the same evidence base, restructured for each audience.

Single-session delivery
What used to take 6–12 weeks of consultant engagement is delivered in one structured session — with full evidence traceability.
Five deliverables, one evidence base
Strategic Summary, Operational Handbook, Medical Writer Handoff, Compliance Extract, and MSL Field Guide — distilled, not regenerated.
Persistent orchestration
Plans live, sleep, and wake up. Programmes go dormant for weeks and reactivate with full strategic memory intact.
Phased MAPS Workflow
Follows a five-phase Medical Affairs Plan of Strategies methodology: context enrichment, landscape analysis, strategic assessment, tactical recommendations, and execution planning.
Live Data Integration
Queries PubMed, ClinicalTrials.gov, EU CTIS, WHO ICTRP, FDA, EMA, NICE, and regional registries across LATAM and Asia-Pacific. MeSH term expansion ensures comprehensive retrieval — nothing relevant is missed because of phrasing.
Session Manager
Persistent session architecture. Tracks context, manages deliverable state, and coordinates multi-agent workflows across extended runs.
Monte Carlo Simulation
Probabilistic scenario modelling for regulatory timelines and competitive launch sequences. Quantifies uncertainty across thousands of simulations.
Desktop-Native, Data-Sovereign
A native desktop application with local session storage. Your data stays on your machine. GDPR-compliant by architecture. Exports directly to Word and PDF.
Compliance Built In
Every output includes source citations, confidence qualifiers, and a compliance footer. Regulatory claims are validated against source agencies.
Symbolic knowledge architecture
At the core of sAÏmone sits a structured knowledge base encoding pharmaceutical Medical Affairs domain logic: strategic pillars, tactical taxonomies, stakeholder profiles, evidence hierarchies, and metric definitions — all with explicit interdependencies, constraint rules, and feasibility gates. Authored, validated, and deterministic.
Cascade contracts & decision space
Cascade contracts link stakeholder scope, tactical proposals, strategic pillars, and evidence tiers into a coherent decision space the substrate reasons within. Stakeholder access shapes which tactics are in play; tactical proposals hold consistency with their parent pillars; claims are bounded by the tier of evidence that supports them. These are authoritative inputs the model is trained to respect — a shaped, inspectable envelope rather than a single chokepoint — which is what keeps the reasoning navigable, domain-appropriate, and defensible.
Evidence epistemology
Every data point carries an explicit verification status: verified against primary sources, inferred from validated data, or flagged for internal confirmation. A four-tier evidence hierarchy governs what each source is allowed to claim — peer-reviewed publications support outcome-level claims; preprints are limited to activity-level observations.
Computational strategy layer
Beyond qualitative frameworks, sAÏmone runs live quantitative analysis within sessions: Monte Carlo simulations for regulatory timeline uncertainty, game-theoretic modelling for competitive scenarios, and decision-theoretic reasoning under incomplete information. Executable computational models, not summaries of theory.
GLP-1 Agonists
Metabolic / CardiovascularADC Oncology
Solid TumoursHIV
Infectious DiseaseNMIBC
Uro-oncologyAtopic Dermatitis
Dermatology / ImmunologyMyasthenia Gravis
Rare Disease — NeurologySMA
Rare Disease — NeurologyAlzheimer's
Neurology+ More
Therapy-agnostic by design
HIV Portugal — Competitive Landscape
Market access, KOL mapping, regulatory status, and evidence positioning across 12 agencies
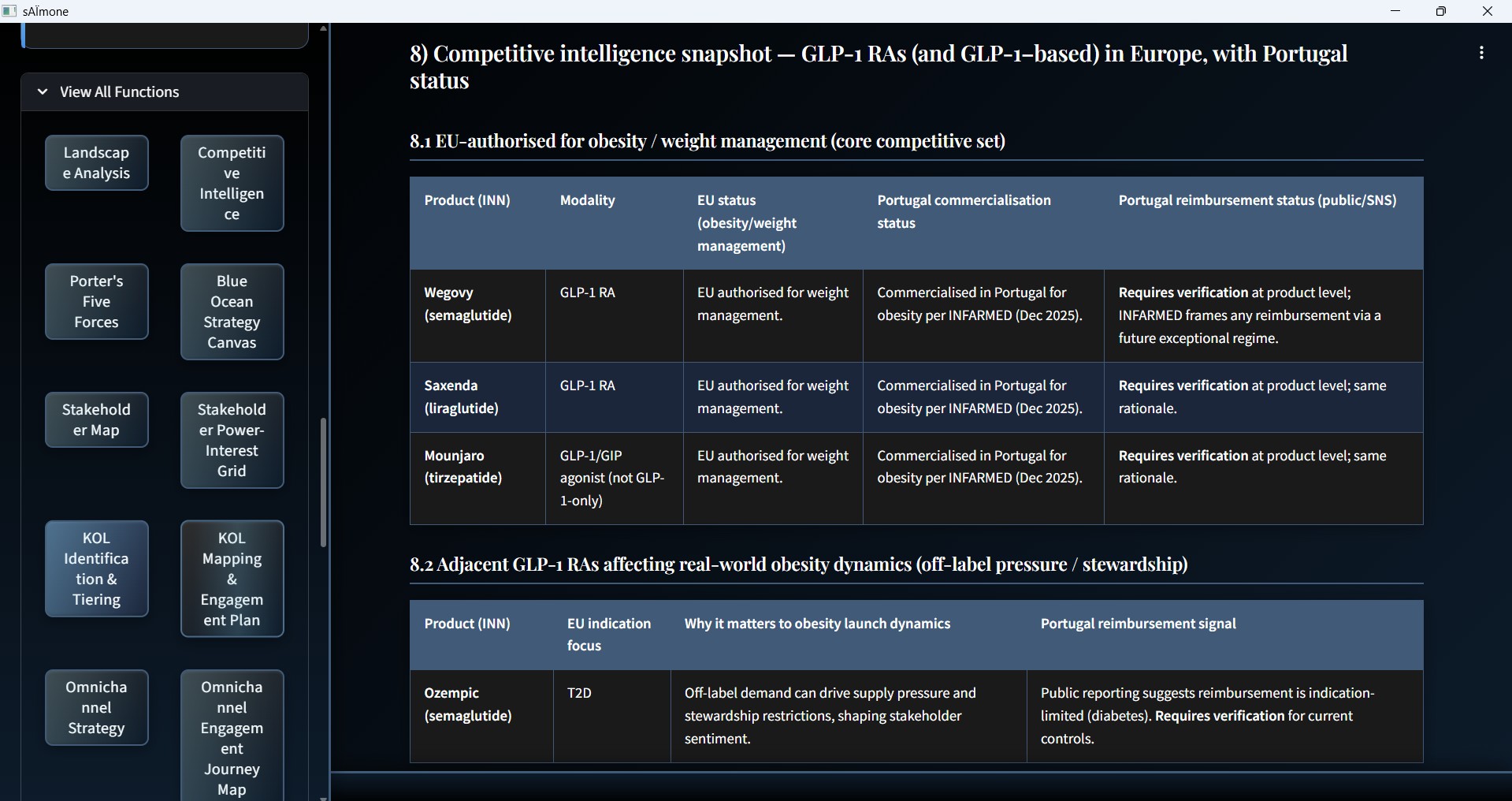
Competitive Intelligence Snapshot
EU-authorised product comparison with modality, regulatory status, and reimbursement by country

Evidence Gap-to-Action Analysis
Evidence gaps mapped to PCIPO priorities with specific Medical Affairs actions for each gap
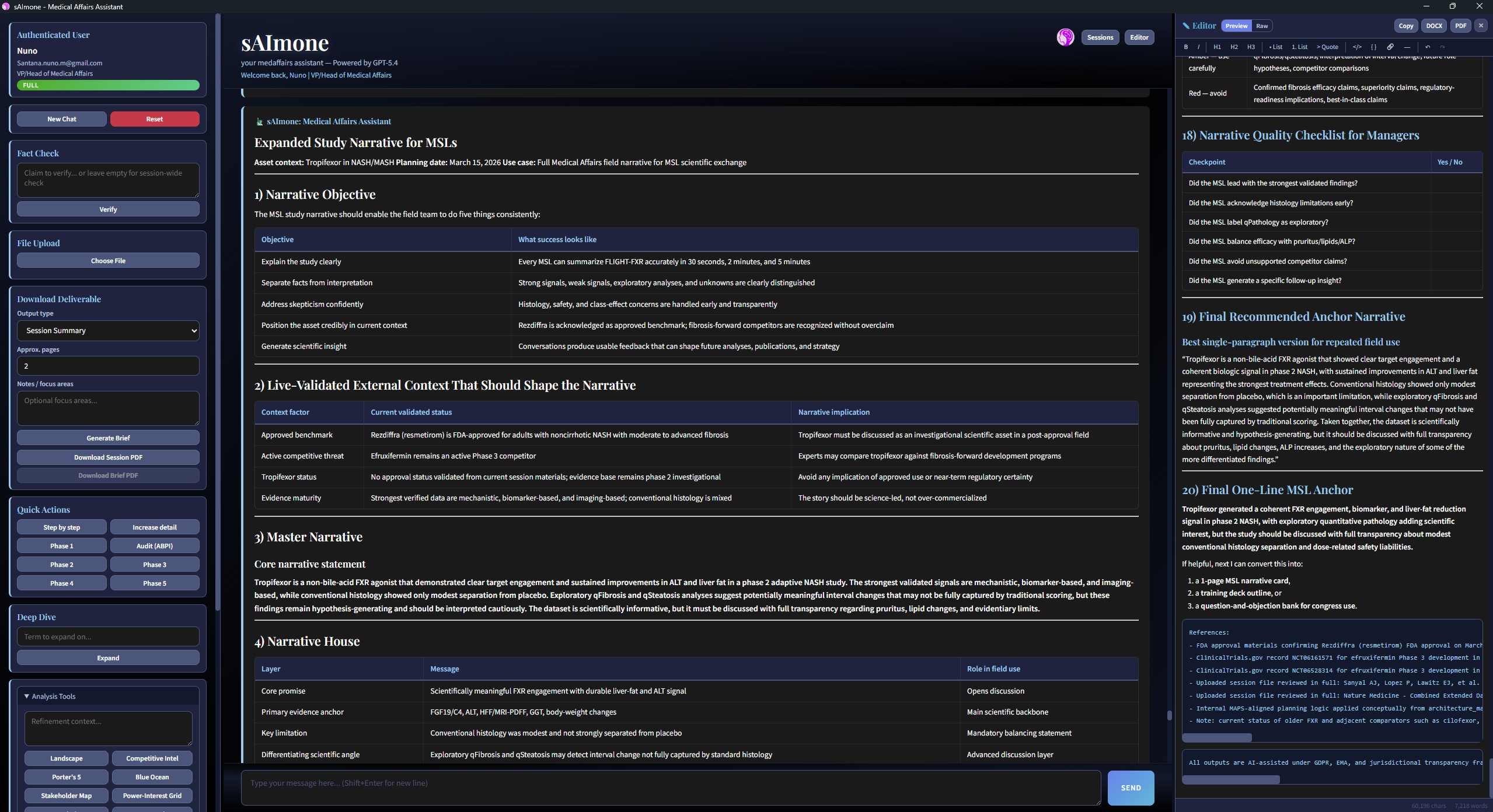
Expanded Study Narrative
Live-validated context, master narrative, quality checklist, and compliance grading
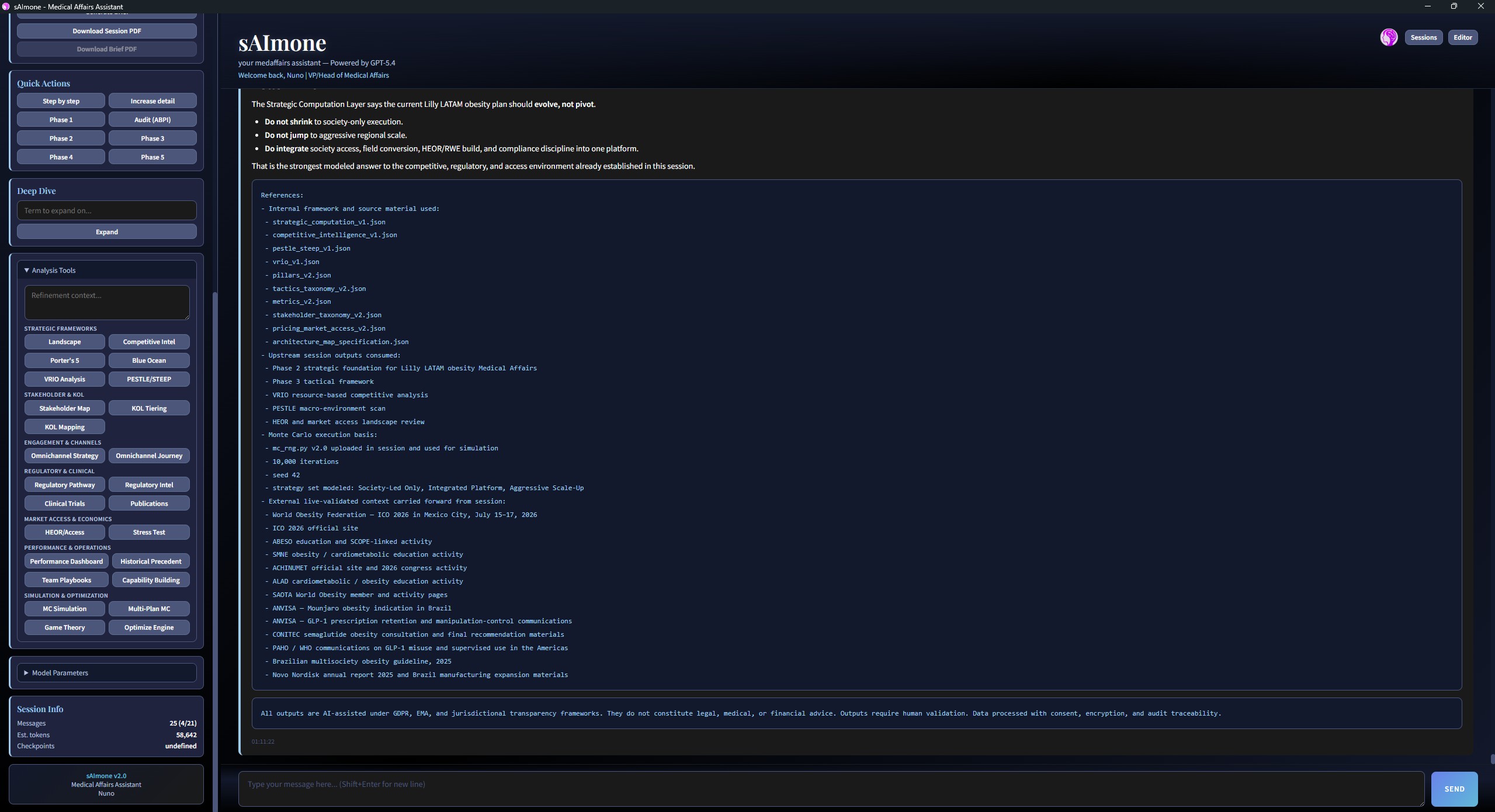
Strategic Computation Layer
Decision-theoretic analysis with assumptions, confidence qualifiers, and inter-model sandbox
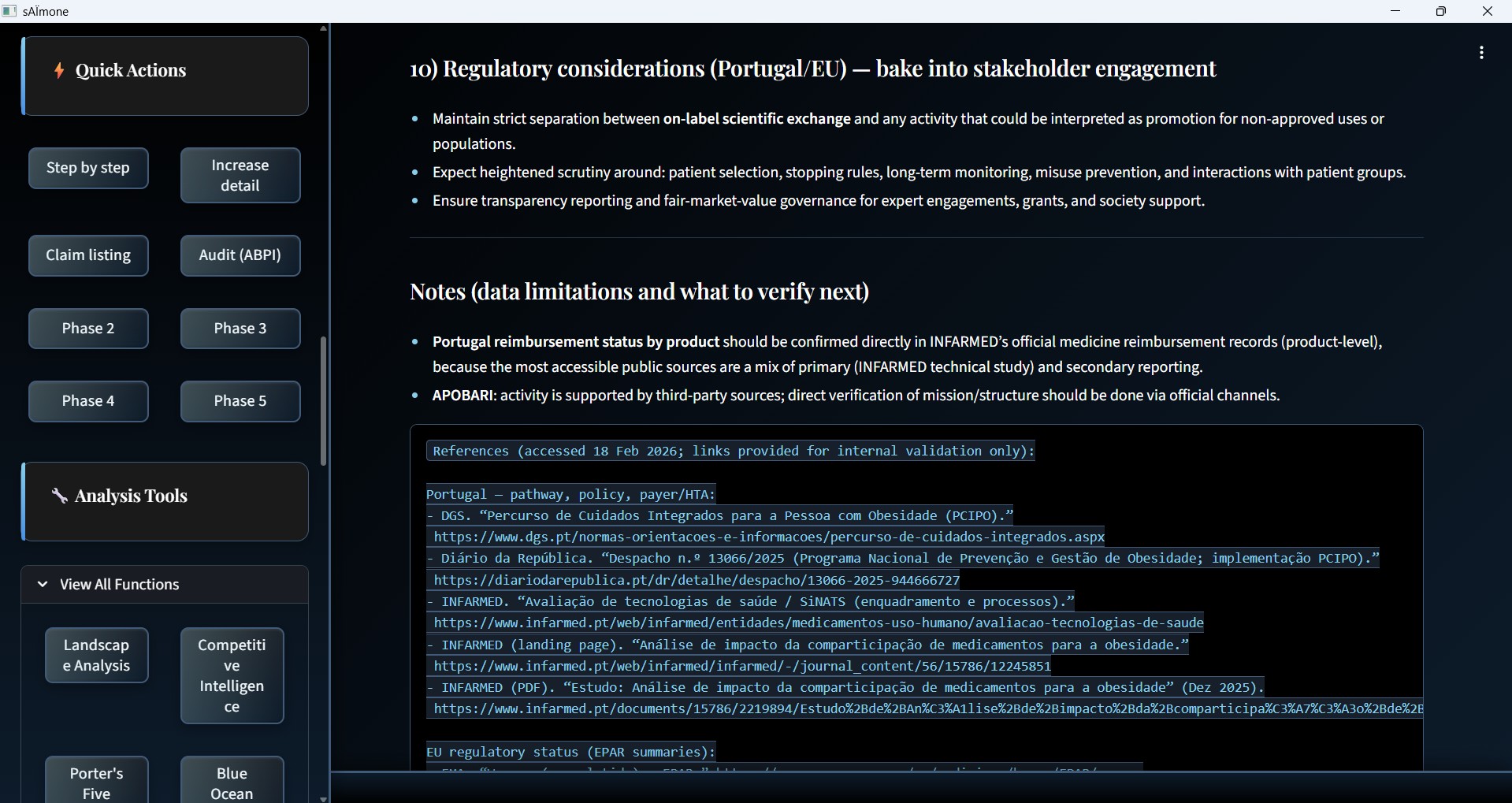
Portugal / EU — Regulatory Considerations
Regulatory access context, reimbursement verification, and data limitations flagged with audit URLs
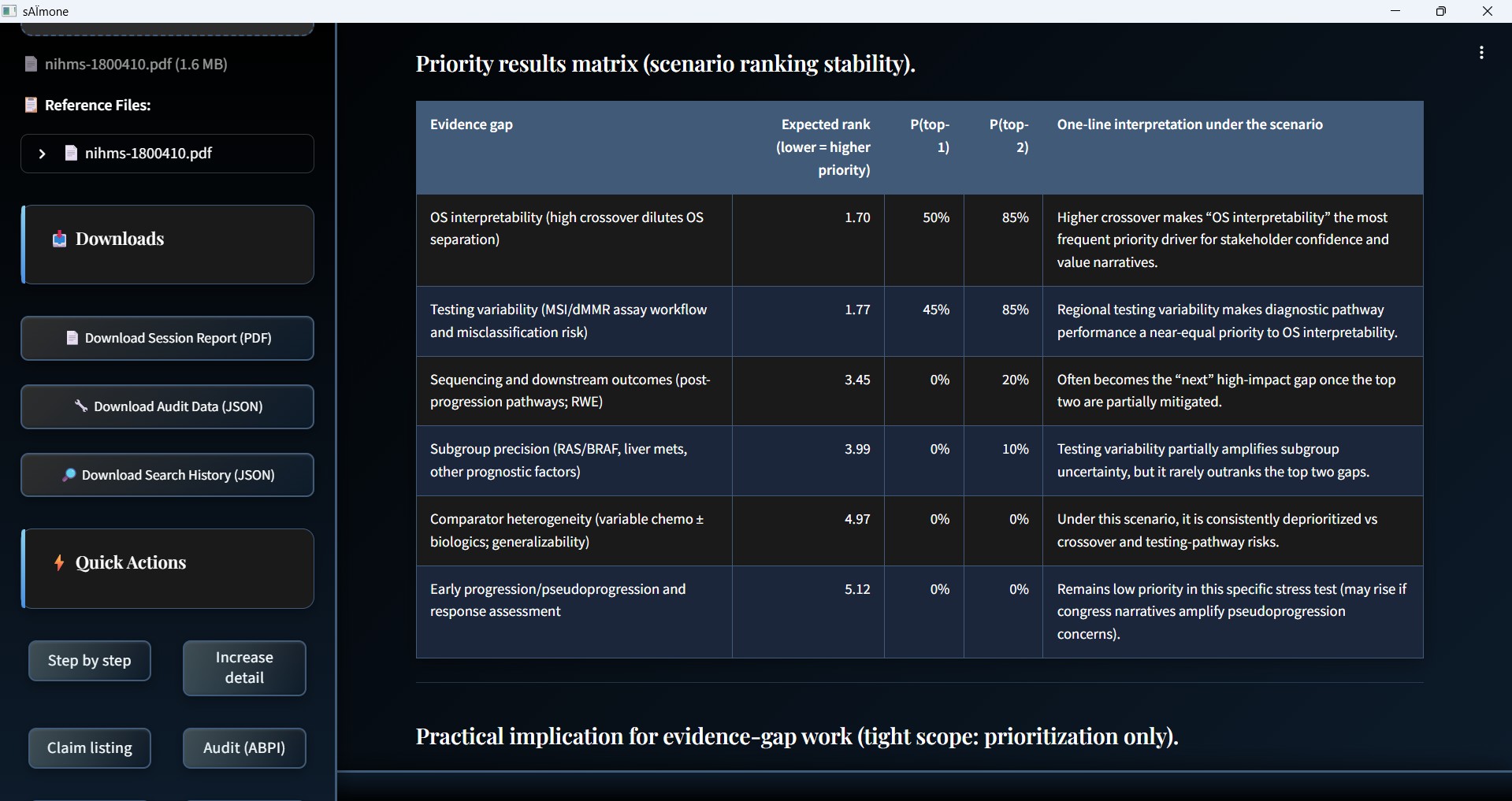
Monte Carlo Simulation Engine
Live computational analysis modelling uncertainty across thousands of scenario iterations
Session Manager
Persistent session architecture — context tracking and multi-agent workflow coordination
sAÏmone is currently available through direct engagements with Medical Affairs teams. We're happy to walk you through the platform and what it produces.
Whether you're exploring AI tools for Medical Affairs, looking to improve your competitive intelligence workflow, or simply want to see a demo — we'd welcome the conversation.
saimone.avti@gmail.com